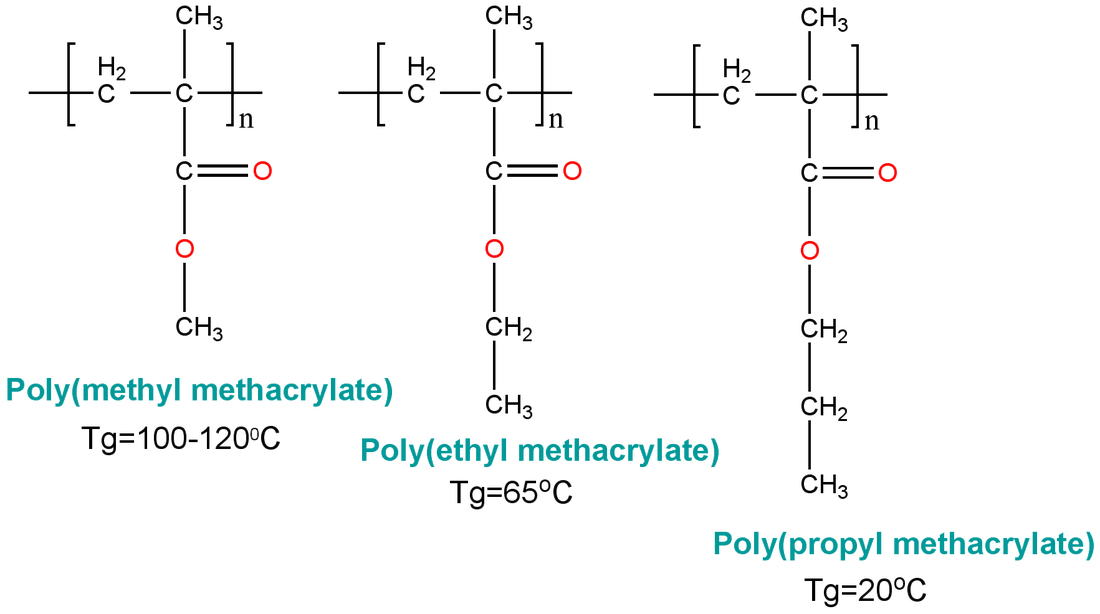
Poly Methyl Methacrylate

Poly methyl methacrylate (PMMA) also referred to as "Acrylic Glass" or "Safety Glass" is a transparent thermoplastic that a lightweight, shatterproof alternative to glass.
The IUPAC name for the polymer of Poly methyl methacrylate is Poly (methyl 2-methylpropenoate).
The Functional group that it contains is an Ester. This can be identified by the
(R Group) - O - C=O - (R Group)
The IUPAC name for the polymer of Poly methyl methacrylate is Poly (methyl 2-methylpropenoate).
The Functional group that it contains is an Ester. This can be identified by the
(R Group) - O - C=O - (R Group)
WHAT IS IT USED FOR?
It is used in products such as plexiglass and other transparent glass substitutes, it is appearing as an ingredient in a number of cosmetics and aestheticians' offices as a filler for wrinkles and fine lines.It's naturally compatible with human tissue, and was a frequent a component of contact lenses in the past; it's also been used for dentures and bone replacement, when combined with bovine collagen.
It can also be used to improve the skin's contour and reduce depression in the skin due to scars, injuries or lines. They are used to correct facial lines and features that could have been treated with collagen replacement thearpy or hyaluronic therapy.
It can also be used to improve the skin's contour and reduce depression in the skin due to scars, injuries or lines. They are used to correct facial lines and features that could have been treated with collagen replacement thearpy or hyaluronic therapy.
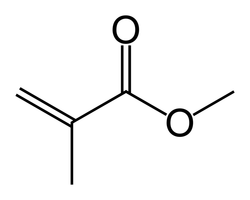
Skeletal structure of methyl methacrylate, the constituent monomer of PMMA. The Ester group here is very easily identified.
PMMA CAN HELP
- Frown lines that run between the eyebrows
- Smoker's lines which are vertical lines on the mouth
- Marionette lines at the corner of the mouth
- Worry lines that run across the forehead
- Crow's feet at the corner of the eyes
- Deep smile lines that run from side of the nose to corners of the mouth
- Redefining lip border
- Acne scars
- Some facial scars
CHEMICAL REACTIONS & HALF-LIFE IN THE ENVIRONMENT
As methyl methacrylate is highly reactive with hydroxyl radicals, its estimated half-life in the troposphere is short; from <5 hours in summer to a few days in winter at a latitude such as that of Toronto, Canada. The reported photo-oxidation half-life of methyl methacrylate is 1.1-9.7 hours. Methyl methacrylate is readily polymerized by light and heat but is not expected to photolyze (Government of Canada, 1993).
In neutral or acidic aquatic environments, hydrolysis of methyl methacrylate is not significant. Based upon its measured second-order hydrolysis rate constant of 200 (mol/litre) at 25oC, the hydrolysis half-life of methyl methacrylate is estimated to be 3.9 years at pH 7 and 14.4 days at pH 9 (Howard, 1989).
Bio-degradation contributes significantly to removal of methyl methacrylate from the environment. The aqueous aerobic degradation half-life is estimated to be 1-4 weeks, and the anaerobic degradation half-life is estimated to be 4-16 weeks (Howard, 1989).
In neutral or acidic aquatic environments, hydrolysis of methyl methacrylate is not significant. Based upon its measured second-order hydrolysis rate constant of 200 (mol/litre) at 25oC, the hydrolysis half-life of methyl methacrylate is estimated to be 3.9 years at pH 7 and 14.4 days at pH 9 (Howard, 1989).
Bio-degradation contributes significantly to removal of methyl methacrylate from the environment. The aqueous aerobic degradation half-life is estimated to be 1-4 weeks, and the anaerobic degradation half-life is estimated to be 4-16 weeks (Howard, 1989).